Advancing Brain Tumor Analysis: The Power of Attention-Gated R2U-Net for Segmentation and Prognosis
Explore how the Attention-Gated R2U-Net model revolutionizes brain tumor segmentation and survival prognosis using advanced AI, enhancing treatment planning and patient outcomes.
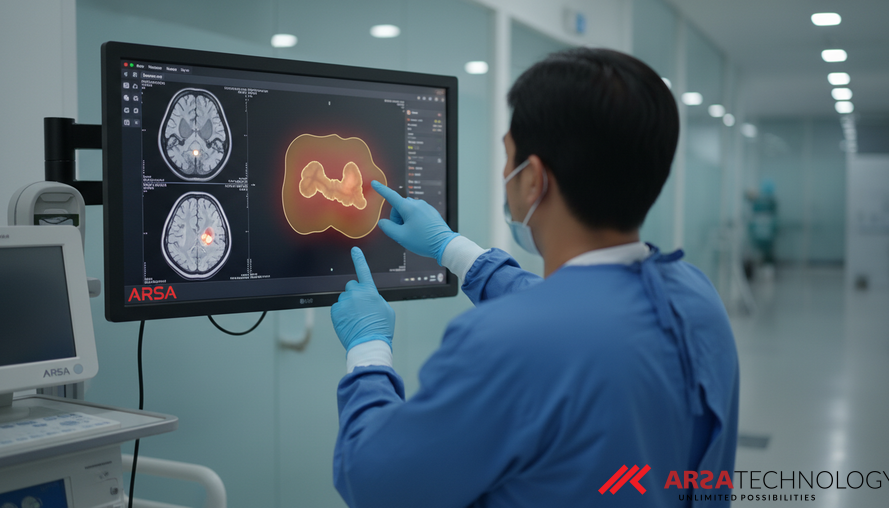
Revolutionizing Brain Tumor Diagnosis with Advanced AI
Brain tumors, particularly gliomas, present a formidable challenge in modern medicine due to their varied aggressiveness, complex characteristics, and demanding treatment protocols. Accurately identifying and characterizing these tumors is critical for effective treatment planning and predicting patient outcomes. Traditionally, manual segmentation of tumors from Magnetic Resonance Imaging (MRI) scans is a time-consuming and often subjective process prone to human error. However, recent advancements in artificial intelligence (AI) are transforming this landscape, offering new avenues for precision and efficiency in medical diagnostics. A recent study by Patel et al. (2026) introduces a novel AI model, the Attention-Gated Recurrent Residual U-Net (R2U-Net) based Triplanar (2.5D) model, designed to significantly enhance brain tumor segmentation and aid in survival prognosis.
The Intricacies of Gliomas: A Medical Imperative
Gliomas are the most prevalent primary brain malignancies, originating from glial cells within the central nervous system. These tumors range from low-grade (LGG) to highly aggressive high-grade gliomas (HGG), often referred to as glioblastomas, which are characterized by their rapid growth and diffusive boundaries. Despite ongoing technological progress, the median survival rate for glioblastoma patients remains critically low, underscoring the urgent need for more precise diagnostic and prognostic tools. The inherent heterogeneity of gliomas, including subregions like edematous lesions, necrotic cores, and active enhancing cores, makes them particularly challenging to delineate and monitor. This complexity is mirrored in their medical imaging, where each subregion exhibits distinct intensity patterns across multiparametric MRI scans, reflecting unique biological properties vital for accurate diagnosis and treatment.
Magnetic Resonance Imaging (MRI) is the gold standard for tumor examination due to its non-invasive nature, high contrast, and detailed resolution. However, the sheer volume and complexity of MRI data make manual analysis by oncologists extremely labor-intensive and susceptible to variability. Automating this segmentation process would be a monumental step forward, providing oncologists with faster, more consistent, and objective insights for early diagnosis and personalized treatment strategies. The Brain Tumor Segmentation (BraTS) challenge provides a benchmark dataset and evaluation platform, defining tasks such as segmenting the Whole Tumor (WT), Enhancing Tumor (ET), and Tumor Core (TC), and importantly, predicting patient survival days (SD), often categorized into long-term, mid-term, and short-term groups.
Introducing the Attention-Gated R2U-Net: A Novel AI Architecture
The core of this innovative approach lies in its sophisticated AI architecture: the Attention-Gated R2U-Net based Triplanar model. This system represents a significant leap in medical image analysis, enhancing both the accuracy and efficiency of tumor segmentation. It is built upon several advanced deep learning concepts, each contributing to its superior performance:
- U-Net Foundation: The U-Net is a widely recognized convolutional neural network architecture specifically designed for biomedical image segmentation. Its unique U-shaped structure, consisting of an encoder to capture context and a decoder to enable precise localization, makes it exceptionally effective at identifying detailed structures within medical images.
- Attention Gates: These mechanisms allow the network to dynamically focus on relevant regions of interest within the image while suppressing irrelevant background information. This selective attention improves the model's ability to delineate tumor boundaries accurately, especially in heterogeneous areas.
- Recurrent and Residual Connections: The "R2" in R2U-Net signifies the integration of recurrent and residual connections. Residual blocks address the "vanishing gradient problem" in deep networks, facilitating the training of very deep models by allowing information to bypass certain layers. Recurrent connections, on the other hand, enable the model to capture temporal relationships and iteratively refine feature representations, leading to a richer and more comprehensive understanding of the tumor structure.
- Triplanar (2.5D) Approach: Instead of processing the entire 3D volume at once (which is computationally intensive), the model uses three identical 2D networks. Each network is trained on different planar views of the MRI scan (sagittal, coronal, and axial slices). The outputs from these three networks are then averaged to produce a final, robust segmentation. This 2.5D approach significantly reduces computational complexity, simplifies data acquisition, minimizes memory requirements, and enhances interpretability without sacrificing accuracy.
This combination of techniques allows the model to deliver highly detailed and context-aware segmentation, crucial for understanding the complex nature of brain tumors. Deploying such advanced custom AI solutions can transform how medical images are analyzed.
Precision Segmentation: Unlocking Detailed Tumor Insights
The effectiveness of any segmentation model is measured by its ability to accurately identify and outline the target regions. The proposed Attention-Gated R2U-Net model demonstrates impressive performance in this regard. On the BraTS2021 validation dataset, the model achieved a Dice Similarity Score (DSC) of 0.900 for Whole Tumor (WT) segmentation. The Dice Similarity Score is a metric that measures the overlap between the AI's predicted segmentation and the actual tumor boundaries, with a score of 1.0 indicating perfect overlap. A score of 0.900 signifies a high degree of accuracy, comparable to leading models in the field.
This high level of precision in delineating the whole tumor, along with its various subregions (Enhancing Tumor and Tumor Core, although specific DSCs for these were not detailed in the provided abstract), offers significant advantages. Accurate segmentation ensures that clinicians have a clear, objective map of the tumor's extent, which is vital for:
- Pre-surgical Planning: Guiding surgeons to remove as much of the tumor as possible while preserving healthy brain tissue.
- Radiation Therapy Planning: Precisely targeting radiation doses to the tumor while minimizing exposure to surrounding critical structures.
- Treatment Response Assessment: Objectively tracking changes in tumor size and composition over time to evaluate the effectiveness of therapies.
Such precise AI video analytics capabilities can be deployed in a variety of healthcare settings, integrating with existing systems to provide actionable insights.
Beyond Segmentation: Predicting Patient Survival
Beyond merely outlining the tumor, the model also offers crucial prognostic capabilities. The triplanar network's encoder is leveraged to extract rich features from the segmented images – 64 features per planar model. These features, which might include details about tumor shape, texture, and intensity patterns, are then fed into an Artificial Neural Network (ANN). The ANN's role is to reduce the dimensionality of these features, distilling them into a more manageable set of 28 key indicators, which are then used to predict patient survival days.
The results for survival prognosis, while still an area of active research, show promising initial outcomes:
- Accuracy: 45.71%
- Mean Squared Error (MSE): 108,318.128
- Spearman Rank Correlation Coefficient (SRC): 0.338
While an accuracy of 45.71% may seem moderate compared to segmentation scores, survival prognosis is inherently a complex prediction task with numerous confounding variables. The Mean Squared Error (MSE) provides a measure of the average squared difference between the predicted and actual survival days, while the Spearman Rank Correlation Coefficient (SRC) indicates the strength and direction of the monotonic relationship between predicted and actual survival ranks. An SRC of 0.338 suggests a noticeable correlation, highlighting the model's potential to provide valuable guidance on patient prognosis. These insights, especially when combined with clinical data, can significantly contribute to personalized patient management strategies.
The Practical Impact of Advanced AI in Healthcare
The development of the Attention-Gated R2U-Net model underscores the profound impact AI can have on healthcare. By offering highly accurate and computationally efficient solutions for brain tumor segmentation and prognosis, this research brings us closer to a future where:
- Diagnosis is Faster and More Consistent: Reducing the burden on medical staff and minimizing subjective errors in analysis.
- Treatment Planning is More Precise: Leading to improved patient outcomes and reduced side effects.
- Prognosis is Personalized: Providing clinicians and patients with data-driven insights to make informed decisions about care pathways.
- Research and Development Accelerate: Offering robust tools for analyzing large datasets and uncovering new correlations.
For enterprises and healthcare providers, integrating such specialized AI can streamline operations, enhance diagnostic confidence, and ultimately elevate patient care standards. This aligns with the broader push towards Industry 4.0 in healthcare, leveraging technologies like AI and IoT for smarter, more connected systems.
Future Directions and the Role of Specialized AI
The study acknowledges the ongoing need for efficient and lightweight segmentation networks, especially as models become deeper and more computationally demanding. The 2.5D approach of the Attention-Gated R2U-Net addresses this by striking a balance between performance and computational resources. Future research will likely focus on refining these models further, exploring more advanced attention mechanisms, integrating multimodal data beyond MRI, and improving the accuracy of survival prognosis predictions.
Specialized AI providers, like ARSA Technology, are at the forefront of deploying production-ready AI and IoT solutions across various industries, including healthcare. While ARSA did not develop the specific research model discussed, its expertise in AI API development and custom AI solutions aligns with the practical application of such advanced computer vision capabilities in real-world scenarios. The findings from research like this continue to inform and inspire the development of robust, deployable systems that deliver measurable impact.
For organizations looking to integrate advanced AI into their healthcare operations or any other sector, understanding these sophisticated technologies is the first step. To explore how AI and IoT solutions can transform your business, we invite you to contact ARSA for a free consultation.
Source: Patel, R., Rajput, S., Raval, M. S., Kapdi, R. A., & Roy, M. (2026). Attention-gated U-Net model for semantic segmentation of brain tumors and feature extraction for survival prognosis. arXiv preprint arXiv:2602.15067.
